Low Estrogen AND Estrogen Dominant?
The Paradox Your Doctor Won’t Explain
There’s a war going on in health communities right now, and estrogen is at the center of it.
On one side, the mainstream position: estrogen is essential for bone density, skin collagen, cardiovascular health, brain function, and vaginal tissue integrity. Postmenopausal women who lose estrogen lose bone at 2-3% per year for several years. Skin collagen content drops roughly 30% within the first 5 years after menopause. The Women’s Health Initiative found that estrogen therapy reduced hip fractures by 34% and total fractures by 24%. Transdermal estradiol preserves bone mineral density at the lumbar spine and femoral neck. These are real effects, documented in large studies.
On the other side, certain health communities treat estrogen as something close to a cellular poison. And they have reasons. In certain stress or inflammatory contexts, estrogen promotes cell proliferation, suppresses mitochondrial respiration, increases free radical production, raises cortisol, depletes glycogen, promotes edema, and drives excitotoxic cell death in the brain. Its tissue effects resemble aging, stress, and radiation injury. Progesterone, the argument goes, is the true bone-protective hormone because it opposes cortisol, the real driver of bone catabolism.
Both sides have evidence. And both sides are partly wrong, because both are treating estrogen as a single thing with a fixed character. The truth is more interesting and more useful than either camp admits.
Estrogen is an endogenous hormone. Your body makes it on purpose. It’s part of the reproductive cycle, part of follicular development, part of the signaling cascade that triggers ovulation. It has genuine physiological roles in maintaining epithelial tissue, supporting mucous membrane integrity, regulating calcium absorption in the gut, maintaining vaginal and urethral tissue, and supporting skin hydration and elasticity. The problems arise from estrogen unopposed by progesterone, estrogen in the wrong metabolic context, or estrogen that can’t be cleared. These are ratio problems and clearance problems. They are not evidence that estrogen itself is inherently toxic at physiological levels in a metabolically healthy body.
A young woman with excellent thyroid function, good progesterone output, a healthy liver, flowing bile, and functional gut clearance handles her estrogen fine. She produces it, uses it, clears it. No accumulation or dominance. The issue arises when the metabolic context degrades and the body can no longer process what it produces.
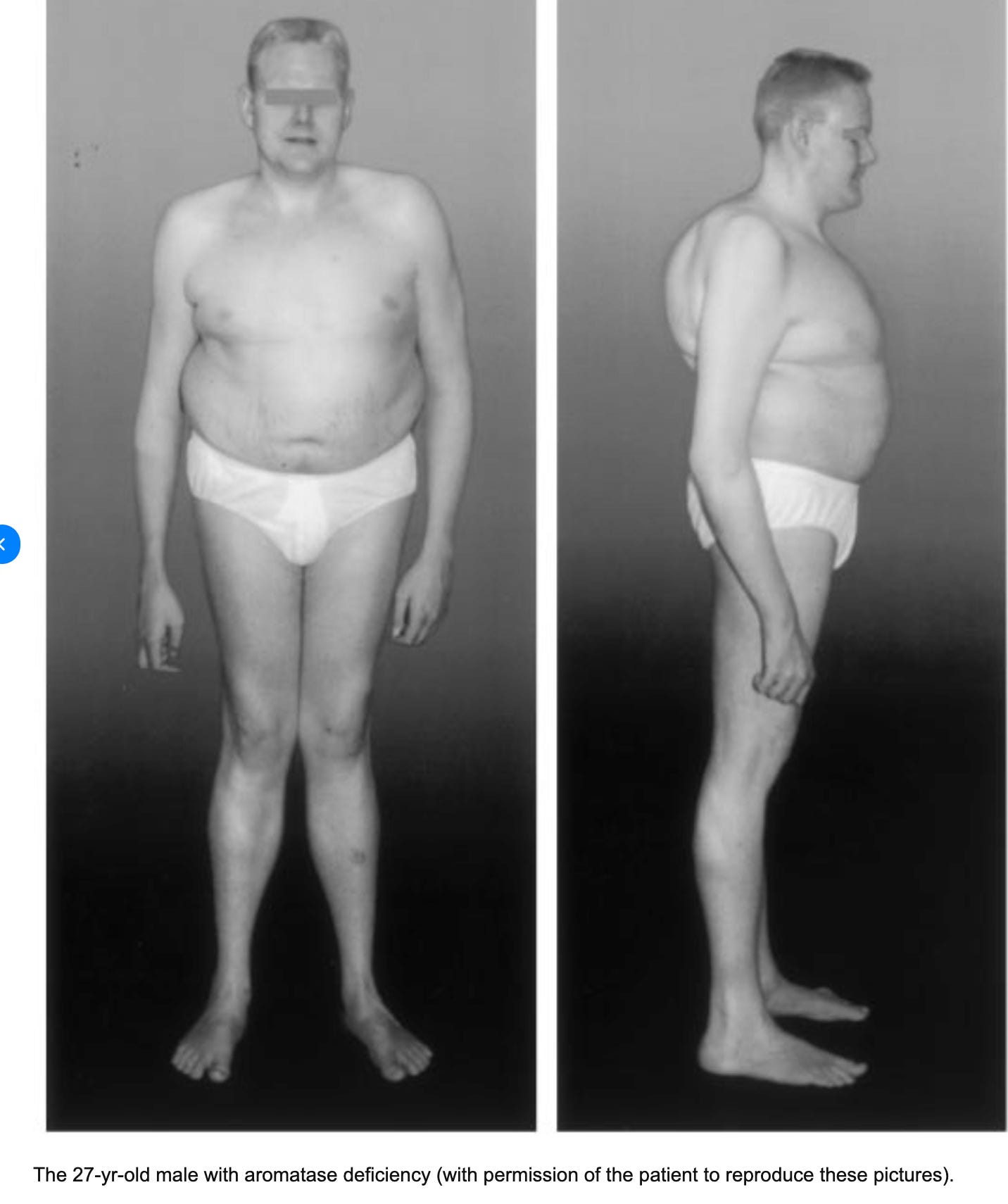
Even men need estrogen. Men with aromatase deficiency, a rare condition in which the enzyme that converts androgens into estrogen is absent or nonfunctional, develop a striking phenotype. They grow abnormally tall because estrogen is what closes the growth plates at the end of puberty, and without it, long bones just keep growing. They develop osteoporosis in their twenties. They get insulin resistance, fatty liver, abnormal lipid profiles, and early atherosclerosis. Their body proportions become eunuchoid, meaning disproportionately long limbs relative to the trunk. These men have plenty of testosterone. They are not lacking androgens. What they lack is estrogen, and their skeleton, metabolism, and cardiovascular system fall apart without it. When given transdermal estradiol replacement, their bone density increases, their epiphyses close, and their insulin resistance improves. This is published in the New England Journal of Medicine, documented across multiple case reports worldwide.
So why do so many women feel terrible from it? Why does estrogen dominance cause so much suffering? And how can a woman be estrogen deficient and estrogen dominant at the same time? Because the problem was never estrogen itself. The problem is context.
Myth #1: Estrogen Dominance Means High Estrogen
This is the biggest misconception. Estrogen dominance was never about absolute estrogen levels. It describes what happens when the normal ratio of estrogen to progesterone is disrupted, either by excess estrogen or inadequate progesterone. The ratio is the signal, and the tissue response follows the ratio.
A woman producing low estrogen can still be estrogen dominant if her progesterone is proportionally lower. A healthy luteal phase requires progesterone to strongly dominate estrogen. In blood measurements, progesterone is typically about 50–200 times higher than estradiol when both are expressed in the same units. When progesterone fails to rise after ovulation, that ratio collapses and estrogen signaling becomes relatively dominant in tissues like the uterus, breasts, pituitary, and brain.
Why does progesterone drop faster than estrogen? Because progesterone production is metabolically expensive. It requires adequate thyroid hormone for the conversion of cholesterol to pregnenolone and downstream to progesterone. It requires stable blood sugar. It requires completed ovulation, because the corpus luteum is where the bulk of progesterone comes from.
Stress, hypothyroidism, poor nutrition, caloric restriction, excessive exercise. All of these suppress progesterone. And when progesterone falls, estrogen goes unopposed. Even small amounts of estrogen can produce dominance symptoms in a progesterone-depleted environment.
The state of estrogen dominance is essentially unstable. The body doesn’t maintain it comfortably. Without adequate progesterone to oppose it, estrogen drives proliferative and inflammatory cascades that feed back into more cortisol, more stress, more estrogen retention. It’s a loop.
Myth #2: If Estrogen Is Low on Labs, You Need More Estrogen
Serum estradiol is a snapshot. A single measurement. It tells you what’s circulating at that moment, which changes throughout the day and dramatically across the menstrual cycle. It does not tell you what’s happening at the tissue level. And it doesn’t tell you how efficiently your body is clearing the estrogen it has.
Plasma estrogen does not reflect tissue concentration of estrogen. Fat cells, the liver, the brain, the uterus can all accumulate estrogen locally through aromatase activity. Aromatase converts androgens to estrogen in peripheral tissues, and its activity is increased by cortisol and decreased by progesterone. So a stressed, hypothyroid, progesterone-depleted woman may have low circulating estrogen and high local tissue estrogen at the same time.
Adding exogenous estrogen in this scenario makes things worse. The tissue exposure increases. The liver burden increases. Progesterone falls further because estrogen suppresses it. Thyroid function takes another hit because estrogen opposes thyroid at multiple levels. The woman feels temporarily different, maybe, because estrogen has anti-cortisol effects in the short term. Then the whole picture degrades.
The question should never be “is estrogen high or low?” alone. The question is: what is the ratio to progesterone, what is the clearance capacity, and what is the metabolic rate?
The Three Chokepoints of Estrogen Clearance
Estrogen leaves the body through a three-stage pipeline. Each stage can fail independently. When any one of them fails, estrogen recirculates regardless of how much was produced in the first place. This is how a woman with low estrogen production ends up functionally estrogen dominant.
Chokepoint 1: Liver Conjugation
The liver metabolizes estrogen through two phases. Phase I (hydroxylation) converts parent estrogens into metabolites via cytochrome P450 enzymes, producing 2-hydroxy, 4-hydroxy, and 16α-hydroxy metabolites. The 2-hydroxy pathway is considered the safest. The 4-hydroxy and 16α pathways produce more reactive, proliferative metabolites.
Phase II (conjugation) takes these metabolites and attaches glucuronic acid, sulfate, or methyl groups to make them water-soluble and excretable. Glucuronidation is the primary route for estrogen elimination.
What impairs liver conjugation? Hypothyroidism slows Phase I and Phase II enzyme activity. Nutrient deficiencies matter here: B vitamins, magnesium, methyl donors like methionine and betaine, glycine for glutathione conjugation. Alcohol competes directly for glucuronidation capacity. Polyunsaturated fatty acids (PUFAs) inhibit glucuronidation.
A sluggish liver treats estrogen like every other toxin it can’t keep up with. It backs up. Estrogen metabolites accumulate. The reactive ones (4-OH, 16α-OH) persist longer than they should.
Chokepoint 2: Biliary Excretion
This one gets overlooked constantly. Even in functional medicine circles, “liver support” is the go-to phrase, as if the liver is where the story ends but it isn’t.
After the liver conjugates estrogen, it packages the conjugated metabolites into bile for delivery to the intestine. Bile is the vehicle. If bile isn’t flowing properly, the conjugated estrogen has nowhere to go. It backs up into circulation and becomes available for reabsorption.
What impairs bile flow? Thick, sluggish bile (cholestasis) can develop when bile production or bile movement slows. Gallbladder dysfunction, bile sludge, or removal of the gallbladder can disrupt the normal release of bile into the intestine. Research shows that women who’ve had cholecystectomy often develop estrogen dominance symptoms months after surgery. Estrogen itself can impair bile flow, and low thyroid function can reduce bile acid synthesis and biliary secretion in the liver. Over time this can create a feedback loop where impaired bile flow reduces the body’s ability to eliminate steroid hormones through bile.
Low-fat diets compound this issue because dietary fat triggers cholecystokinin, the hormone that signals the gallbladder to contract and release bile. Without adequate fat intake, bile sits and thickens.
This is a separate, independent chokepoint. A woman with perfect liver Phase II metabolism still recirculates estrogen if her bile isn’t moving.
Chokepoint 3: Gut Elimination
Bile delivers conjugated estrogen to the intestine. The goal is excretion through stool. Simple enough. Except the gut has its own agenda.
Certain bacteria produce an enzyme called beta-glucuronidase. This enzyme strips the glucuronic acid tag off the conjugated estrogen, reactivating it. The now-unconjugated, biologically active estrogen gets reabsorbed through the intestinal mucosa into the portal vein and re-enters circulation. This is enterohepatic recirculation.
Research using radiolabeled estrogen has shown that roughly 65% of estradiol and 48% of estrone injected intravenously can be recovered in bile, and the majority of deconjugated estrogens in the gut are reabsorbed rather than excreted. The estrobolome, the collection of gut bacteria capable of metabolizing estrogen, is now recognized as a significant regulator of systemic estrogen levels.
High beta-glucuronidase activity means more estrogen gets recycled back into circulation. Gut dysbiosis, small intestinal bacterial overgrowth, chronic constipation (which gives bacteria more time to deconjugate), and antibiotic-disrupted flora all contribute.
So three completely independent systems can each independently cause estrogen recirculation: impaired liver conjugation, impaired bile flow, and impaired gut elimination. A woman can have one, two, or all three working against her simultaneously.
The Metabolic Trap: How Hypothyroidism Ties It All Together
Hypothyroidism is the thread that runs through every chokepoint. Thyroid hormone influences progesterone synthesis, liver detoxification speed, bile acid production and flow, and intestinal motility. A hypothyroid woman is set up for estrogen dominance at every level.
Thyroid hormone drives the conversion of cholesterol to pregnenolone, the precursor to progesterone. Without adequate thyroid, this conversion stalls. Progesterone output drops. The ratio shifts toward estrogen dominance even if estrogen production also falls.
The liver needs thyroid hormone for adequate Phase I and Phase II enzyme activity. Hypothyroid livers are sluggish livers. Conjugation slows down. Estrogen metabolites accumulate.
Thyroid influences bile composition and viscosity. Hypothyroid bile is thick. It doesn’t flow well. It pools instead of moving conjugated estrogen into the intestine.
And hypothyroidism slows gut motility, causing constipation. Longer transit time means more exposure of conjugated estrogen to bacterial beta-glucuronidase. More deconjugation. More reabsorption.
Estrogen, in turn, suppresses thyroid function. It increases thyroid-binding globulin (TBG), which binds up free thyroid hormones and reduces their availability. It competes with thyroid at the receptor level. So estrogen excess worsens hypothyroidism, and hypothyroidism worsens estrogen excess. The loop locks into place.
This is the self-reinforcing loop: estrogen accumulates in a low-metabolic state. Cortisol rises. Progesterone falls. The liver can’t clear the estrogen. The estrogen suppresses thyroid. The low thyroid prevents the body from making progesterone. Round and round.
The Unstable Cycling Pattern
When progesterone is insufficient, ovulation becomes incomplete or absent. Follicles may develop partially, produce erratic bursts of estrogen, but fail to complete the luteal transition. So you get irregular estrogen spikes with no progesterone wave following.
This is the clinical picture: irregular cycles, spotting, heavy periods alternating with light periods, PMS symptoms that vary wildly month to month. The woman’s estrogen is technically low on average but functionally dominant because progesterone is even lower and clearance is impaired.
The erratic pattern itself creates more stress. Cortisol rises to compensate for the unstable hormonal environment. Cortisol increases aromatase activity in peripheral tissues, converting more androgens to estrogen locally. Cortisol also competes with progesterone for receptor binding. More cortisol means less functional progesterone, even if serum levels look passable.
This is why measuring hormones on a single day often misses the picture entirely. The problem is dynamic. It’s a pattern of instability, a ratio problem, and a clearance problem all layered on top of each other.
Standard Testing Misleads
Serum estradiol is a snapshot. One moment in time. Estradiol fluctuates throughout the day and changes dramatically across the menstrual cycle. A draw on day 3 looks completely different from day 14 or day 21. A single number cannot capture a dynamic, cycling system.
Serum doesn’t measure metabolites. It tells you how much estradiol is circulating right now, but it doesn’t tell you whether your body is clearing it through safe pathways (2-hydroxy) or problematic ones (4-hydroxy, 16α-hydroxy). Two women with identical serum estradiol can have completely different metabolite profiles and completely different risk profiles.
Serum doesn’t reflect tissue concentration. Aromatase in fat cells, the brain, the liver, and other tissues converts androgens to estrogen locally. A woman can have low circulating estradiol and high local tissue estrogen at the same time. Plasma estrogen does not equal tissue estrogen.
And serum doesn’t show you the ratio. An estradiol level of 60 pg/mL means something very different when progesterone is 20 ng/mL versus when progesterone is 0.5 ng/mL. Without seeing both numbers together, the estradiol number alone is nearly meaningless.
The Symptom Distinction: Deficiency vs. Dominance… or Both??
True estrogen deficiency and estrogen dominance are often presented as two separate conditions with clean, distinct symptom profiles. Reality is messier than that. There are patterns associated with each, but they frequently coexist in the same woman. Understanding what each pattern looks like still matters, because it helps you identify which layers of the problem are active.
Symptoms that point toward genuine estrogen insufficiency at the tissue level tend to involve drying and thinning. Vaginal dryness and atrophy. Painful intercourse. Recurrent urinary tract infections, because estrogen maintains the vaginal and urethral epithelium, and without it the tissue thins, pH rises above 4.5, and protective lactobacilli decline. Skin that loses elasticity and wrinkles faster. Joint stiffness and dryness, sometimes mistaken for early arthritis. Accelerated bone loss. Classic vasomotor hot flashes, the kind that wake you up drenched at 3 AM. Shrinking breast tissue.
Symptoms that point toward too much estrogen relative to progesterone, or estrogen that’s recirculating instead of being cleared: swelling, proliferation, fluid retention. Breast tenderness, bloating, especially premenstrually. Heavy periods with clotting. Weight gain concentrated in the hips, thighs, and midsection. Water retention in the hands and face. Mood swings, irritability, weepiness, anxiety. Fibroids, endometriosis, ovarian cysts.
Hot flashes and mood changes can appear in either picture. Unstable estrogen, meaning erratic spikes and drops from incomplete ovulation, can produce hot flashes even in a woman who is functionally estrogen dominant. So hot flashes alone confirm nothing. You need the broader pattern.
A woman who is both low in estrogen production and functionally estrogen dominant may experience symptoms from both lists simultaneously. Vaginal dryness and breast tenderness. Thinning skin and heavy periods. Joint stiffness and bloating. That mixed presentation is itself the diagnostic signal. It means her ovaries aren’t producing enough estrogen (production deficiency), her progesterone has dropped even further (ratio problem), and her liver, bile, or gut can’t clear the estrogen that does exist (clearance problem). She is simultaneously deficient in production and dominant in tissue effect.
This is where the real danger lives. If a practitioner only sees the deficiency symptoms and gives her estrogen, the dominance worsens. If they only see the dominance symptoms and start clearing estrogen aggressively with DIM or calcium D-glucarate, the deficiency deepens. The mixed picture demands a layered approach, and recognizing it for what it is requires looking at the whole system instead of picking one column and running with it.
Better Testing Approaches
If you want lab confirmation beyond symptoms, here’s what actually gives you useful information.
The DUTCH test (Dried Urine Test for Comprehensive Hormones) is currently the most informative single panel available. It measures not just estrogen levels but estrogen metabolites across all three Phase I pathways: 2-hydroxy (generally protective), 4-hydroxy (more reactive, potentially genotoxic), and 16α-hydroxy (proliferative). This shows you how your body is processing estrogen, which is as important as how much you have. It also measures the progesterone-to-estrogen ratio, cortisol and cortisone patterns across the day, DHEA, melatonin, and organic acid markers that reveal B6, B12, and glutathione status. These nutritional markers directly relate to methylation and conjugation capacity, telling you whether your liver has the cofactors it needs to clear estrogen properly. For cycling women, the DUTCH Cycle Mapping option lets you track estrogen and progesterone metabolites across an entire menstrual cycle rather than a single day, which reveals patterns that spot testing will always miss. That said, DUTCH has limitations: it cannot measure FSH or LH, it’s not FDA-cleared, and results require a practitioner who understands metabolite interpretation. I’ve written a separate deep-dive on the strengths and limitations of DUTCH testing for those who want more detail.
Serum testing still has a role. FSH and LH can only be measured in blood, and DUTCH can’t capture them. Elevated FSH with low estradiol is the clearest marker of genuine ovarian insufficiency or approaching menopause. High FSH means the pituitary is screaming at the ovaries to produce estrogen and they’re not responding. This is the most reliable indicator that true production deficiency exists. Combining serum FSH/LH and estradiol with a DUTCH metabolite panel gives the fullest picture: production status from blood, clearance and metabolism from urine.
For cycling women, timing matters. Serum estradiol and progesterone should ideally be drawn on day 19-22 of a 28-day cycle (the mid-luteal phase) to capture peak progesterone. Drawing on day 3 tells you about baseline estrogen but misses the progesterone picture entirely.
Stool testing for beta-glucuronidase levels (available through GI-MAP and similar comprehensive stool panels) tells you directly whether gut bacteria are deconjugating estrogen and sending it back into circulation. Elevated beta-glucuronidase on a stool test points specifically to the gut clearance chokepoint.
Functional Markers You Can Track at Home
A pro-metabolic approach to assessment is practical and low-tech, and it’s still useful.
Basal body temperature and resting pulse. Waking temperature consistently below 97.8°F and resting pulse below 70 bpm suggest insufficient thyroid function, which as we’ve covered is the thread connecting every chokepoint. A healthy metabolic rate produces warmth and a steady heart rate. If your temps are low and your pulse is sluggish, your thyroid isn’t driving the system hard enough to clear estrogen, produce progesterone, or move bile.
Vaginal pH. You can test this with simple pH strips. Healthy, estrogen-supported vaginal tissue maintains a pH below 4.5 because estrogen promotes glycogen production in vaginal epithelium, which feeds lactobacilli, which produce lactic acid. A pH consistently above 4.5, especially with symptoms of dryness and tissue thinning, is a functional marker of insufficient estrogen at the tissue level.
Cycle tracking. Not just period dates, but symptom patterns across the full month. Basal body temperature charted daily can show whether you’re ovulating (temperature shifts up after ovulation due to progesterone’s thermogenic effect) and whether luteal phase temperatures stay elevated for at least 10-12 days. A short, weak luteal phase with temperatures that barely rise or drop quickly confirms insufficient progesterone.
Bowel transit time. How long it takes food to move through you matters for estrogen clearance. If you’re constipated (going less than once daily, or stools are hard and dry), your gut has more time to deconjugate and reabsorb estrogen. You can test transit time by eating a handful of raw beets or activated charcoal and tracking how long it takes to see the color change in your stool. Twelve to twenty-four hours is ideal. Longer than that means estrogen has extra time to recirculate.
The Key Question Before Any Intervention
Before you supplement anything, before you add progesterone or estrogen or calcium D-glucarate etc., you need to answer one question clearly: are my symptoms caused by too little estrogen production, by too much estrogen relative to progesterone (a ratio problem), or by estrogen that’s being produced and recirculated instead of cleared (a clearance problem)?
True production deficiency may eventually warrant bioidentical estrogen replacement (transdermally, with progesterone, after metabolic foundations are in place). A ratio problem needs progesterone support and thyroid optimization. A clearance problem needs liver, bile, and gut work. When all three coexist, the sequence matters: fix thyroid and metabolic foundations first, support progesterone second, address clearance third, and only then consider estrogen replacement if true deficiency persists after the other layers are corrected. Many women find that once thyroid, progesterone, and clearance are addressed, estrogen production recovers on its own and replacement becomes unnecessary. Applying the wrong solution to the wrong problem doesn’t just fail, it actively makes things worse. Adding estrogen to a woman with impaired clearance increases her toxic load!
The Delivery Problem: Not All Estrogen Therapy Is Equal
The estrogen debate is further muddied because most of the negative outcomes from HRT studies involved oral conjugated equine estrogens (Premarin), which are not bioidentical and undergo extensive first-pass liver metabolism. When you swallow estrogen, approximately 95% gets converted to estrone and other metabolites on first pass through the liver. This creates supraphysiologic estrogen exposure in hepatic tissue, increases C-reactive protein, raises clotting factors, increases sex hormone binding globulin, and promotes gallstone formation. These are delivery-route problems, not estrogen-per-se problems.
Transdermal estradiol bypasses the liver entirely. It enters systemic circulation directly, maintaining a more physiological estradiol-to-estrone ratio (roughly 1:1, similar to what ovaries produce). Research consistently shows that transdermal delivery avoids the increased clotting risk associated with oral estrogen. It avoids the inflammatory marker elevation. It avoids the gallbladder complications. It provides bone protection, symptom relief, and tissue support at doses 20 times lower than oral equivalents.
This distinction matters enormously. A woman taking oral Premarin and a woman using a low-dose transdermal estradiol patch are having fundamentally different biochemical experiences. Lumping them together as “estrogen therapy” obscures everything useful about the conversation.
The type of progestogen matters too. Most negative HRT outcomes involved medroxyprogesterone acetate (MPA), a synthetic progestin that has anti-progesterone effects and doesn’t behave like bioidentical progesterone at all. Combining oral equine estrogens with a synthetic progestin is a completely different intervention than using transdermal bioidentical estradiol with bioidentical progesterone. The former has documented risks. The latter has a dramatically different safety profile.
Looking at the Whole System
The physiology here is complex, and anyone claiming to have a single universal answer for estrogen problems is oversimplifying the biology.
What we can say with reasonable confidence: estrogen dominance, meaning an unfavorable ratio of estrogen to progesterone combined with impaired clearance, is a real and common problem that causes real symptoms. Progesterone is profoundly undervalued in mainstream medicine and deserves far more attention. Most women need more progesterone support, not more estrogen.
And also: some women genuinely benefit from estrogen supplementation when true deficiency exists, especially postmenopausally, especially for bone and tissue maintenance, especially when delivered transdermally as bioidentical estradiol alongside adequate progesterone. The key is that estrogen replacement should only be considered when deficiency is confirmed and the metabolic infrastructure to handle it is in place. Thyroid function must be adequate. Progesterone must be sufficient to oppose it. Liver clearance and bile flow must be functional. Without those conditions met first, adding estrogen simply adds fuel to the fire.
Critics of estrogen are correct about several things. Progesterone is the stabilizing hormone in the female system, and estrogen does tend to accumulate in low-metabolic states where progesterone production, thyroid function, and clearance mechanisms are impaired. They are also right that the pharmaceutical history of estrogen therapy created real harm, largely because non-physiological formulations and delivery methods were widely used. But the leap from “estrogen is often problematic” to “estrogen is always a poison” ignores basic physiology. Your body didn’t evolve to produce a toxin. It evolved to produce a hormone that requires specific metabolic conditions to be safe. When those conditions are met, estrogen does its job and leaves. When they aren’t, it accumulates and causes damage.
I’ll share something personal here. In my mid-thirties, I ran both DUTCH testing and daily at-home quantitative urinary hormone monitoring across full cycles, tracking estrogen, LH, and progesterone metabolites day by day. The data showed that my estrogen was genuinely slow to pick up and consistently below range. I had unstable ovulation. I experimented with transdermal estrogen patches and bio-identical progesterone. Eventually, I resolved the underlying problem through correcting my eating patterns, bile issues and metabolic foundations, and my estrogen and progesterone stabilized on its own. I didn’t need the patches forever. But I needed them for a period of time. The data told me I had a real deficiency. I addressed it while working on the root cause. That’s the approach this article advocates: test properly, act on what you find, and always work toward metabolic restoration as the long-term solution.
Context determines whether estrogen is protective or destructive.




Very interesting. I am on HRT and have had a hysterectomy with both ovaries removed. Recently having issues of excess breast tissue growth. Some of this article sounds familiar to what I’ve been going through and praying for an answer. This article mostly addresses those who still have menstrual cycles. But I think things got worse when my gall bladder was removed. Would love more information (or be pointed to where I can find information) about after total hysterectomy and HRT. Is it still important for progesterone to work with estrogen even though no uterus? I’ve always felt better with HRT, but with recent problems wondering about going off it. I find it so confusing and feel like I don’t really get the information I need from any doctor I’ve seen.
Such an important and informative article. Thank you.