The Cancer-Fighting Organ Most People Have Never Heard Of
Surgeons routinely removed it during heart surgery. Patients paid for it with their lives. It sits at the center of your immune system and shrinks with age.
There’s an organ sitting behind your breastbone, right above your heart, that most people have never heard of. The ancient Greeks thought it housed the soul. Modern medicine mostly ignores it. Its called the thymus.
In a newborn, it weighs about 15 grams. By puberty, it reaches its peak, sometimes 40 to 50 grams. Then it begins shrinking. By age 40, roughly 70% of the thymus has been replaced by fat. By 60, it can be hard to find on imaging at all.
For decades, the medical establishment treated this shrinkage as harmless, a relic organ losing relevance after childhood. That assumption turned out to be spectacularly wrong.
What does the thymus actually do?
The thymus is where your immune system learns to tell the difference between you and everything that wants to kill you.
Bone marrow produces immature white blood cells called thymocytes. These cells migrate through the bloodstream into the thymus, where they undergo the most rigorous training program in human biology. The “T” in T-cell stands for thymus. Every T-cell you have passed through this gland.
Inside the thymus, thymocytes go through two selection processes. First, positive selection: can this cell recognize the body’s own tissue markers (MHC molecules)? If yes, it moves forward. If no, it dies. Then comes negative selection: does this cell attack the body’s own proteins? If yes, it dies. If no, it graduates and enters circulation.
Here’s what makes this remarkable. About 98% of developing thymocytes fail these tests and are destroyed inside the thymus through programmed cell death. Only 2% survive. The thymus kills its own students at a rate that would make any drill sergeant blanch, and for good reason. A T-cell that can’t recognize threats is useless. A T-cell that attacks your own tissues creates autoimmune disease.
The survivors become three main types of fighters:
Cytotoxic T-cells (CD8+) directly kill cells infected by viruses or bacteria. They also kill cancer cells.
Helper T-cells (CD4+) coordinate the immune response. They activate B-cells to make antibodies, prime other T-cells, and direct the whole operation.
Regulatory T-cells suppress immune responses that have gone too far, preventing your own immune system from destroying healthy tissue.
Without the thymus, none of this works. Infants born without a thymus (DiGeorge syndrome) have severe immunodeficiency and often don’t survive without thymus transplantation. Remove the thymus from a newborn mouse, and it dies of infection.
The Thymus as an Endocrine Organ
Most people, including many doctors, think of the thymus only as an immune organ. It’s also an endocrine gland. It produces several hormones that reach far beyond T-cell education.
Thymulin requires zinc to be biologically active. It circulates in the blood and helps mature T-cells after they leave the thymus. When zinc levels drop, thymulin activity drops with it, which partly explains why zinc deficiency so profoundly suppresses immunity.
Thymosin alpha-1 activates dendritic cells, natural killer cells, and T-cells simultaneously, so it strengthens both your first rapid response to a pathogen and the slower, targeted response that remembers that specific pathogen for next time.
Thymopoietin signals the pituitary gland to release hormones, connecting the thymus to the broader hormonal system.
The thymus also produces small amounts of hormones usually associated with other glands: melatonin, insulin, growth hormone analogs, and prolactin. The significance of this extra-thymic hormone production isn’t fully understood, but it hints at a more integrated role in metabolism and aging than we ever thought.
One particularly striking finding: thymulin levels in the blood correlate directly with thyroid hormone levels. Hypothyroid patients have lower thymulin. Hyperthyroid patients have higher thymulin. This isn’t a coincidence. T3 acts directly on thymic epithelial cells, the cells responsible for making thymic hormones. The thymus and thyroid are deeply intertwined.
The 2023 Study
For years, surgeons routinely removed the thymus during cardiac surgery to get better access to the heart. Nobody thought twice about it. The thymus was considered expendable in adults.
Then a 2023 study published in the New England Journal of Medicine blew that assumption apart.
Researchers at Massachusetts General Hospital tracked 1,146 adults who had their thymus removed during surgery and compared them to 1,146 matched controls who had similar surgery but kept their thymus. The results were stark.
At five years, the thymectomy group had nearly triple the all-cause mortality. Their cancer risk doubled. When patients with no prior infections, cancer, or autoimmune disease were analyzed separately, the thymectomy group also had significantly higher rates of new autoimmune disease.
The researchers also measured T-cell production and inflammatory markers in a subset of patients. The thymectomy group produced fewer new T-cells and had elevated inflammatory cytokines, a profile associated with accelerated aging.
A follow-up study confirmed the cancer finding using a broader control group of over 62,000 general medicine patients at the same hospital, not just surgical patients. Adults who lost their thymus had higher rates of cancer death. The thymus, it turns out, is still doing something essential in adulthood: immunosurveillance against cancer.
This has massive implications. It means the gradual involution of the thymus during aging isn’t just an irrelevant process. It’s likely contributing to the rise in cancer, autoimmune disease, and infection susceptibility that comes with getting older.
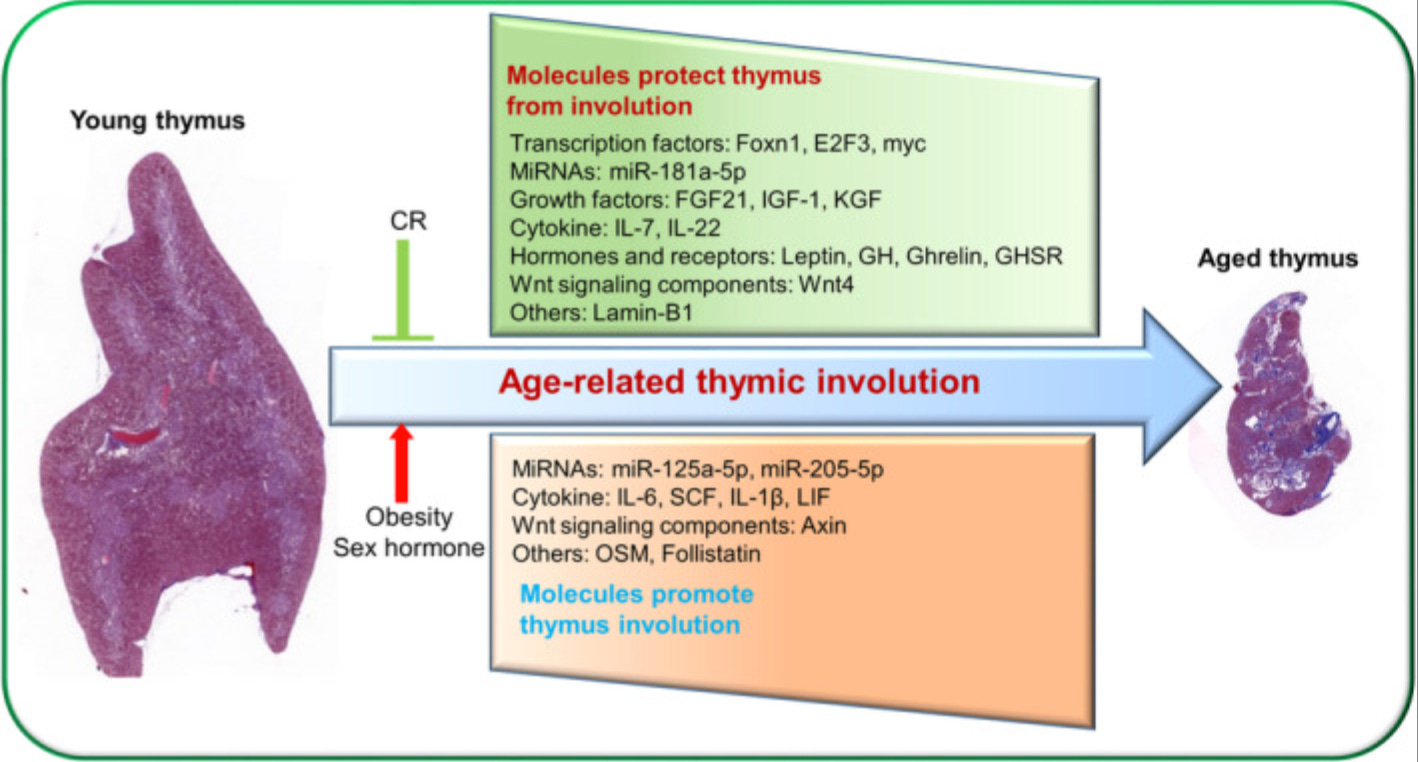
Why the Thymus Shrinks: The Standard Story and What’s Missing
The conventional explanation goes like this: after puberty, rising sex hormone levels trigger the thymus to involute. Growth hormone declines. The thymus shrinks. It’s genetically programmed. End of story.
There’s truth here, but the picture is far more complicated.
Both estrogen and testosterone do promote thymic involution. Castration in rodents causes the thymus to regrow significantly, and administering sex steroids reverses that regrowth. This is consistent and well-documented.
But here’s a finding that complicates the neat narrative: one study using hypogonadal mice (which never produce normal levels of sex steroids) found that their thymus still involuted at the same rate as normal mice. And the thymic regrowth from castration turned out to be transient. The thymus eventually shrank again regardless.
This suggests sex hormones accelerate involution but aren’t the fundamental cause. Something else is driving the process. Several factors have been identified, and they all connect back to the same theme: the metabolic environment around the thymus deteriorates, and the thymus deteriorates with it.
1. The thymus converts itself into fat. By age 40, roughly 80% of the thymic space is occupied by fat tissue. For a long time, researchers assumed fat cells migrated in from outside and crowded out functional tissue. The actual mechanism is different. The thymus’s own structural cells, the epithelial cells that create the environment T-cells need to mature, gradually transform into fibroblasts and then into fat cells. The organ is remodeling itself from the inside out. And these fat cells aren’t passive filler. They actively produce inflammatory signals and steroids that suppress T-cell development, creating a loop: more fat means more inflammation, which means fewer functional cells, which means more fat.
Some degree of this conversion appears to happen in every species studied, so it’s partly biological aging. But the speed is not fixed. Obesity, chronic inflammation, hypothyroidism, high cortisol, and zinc deficiency all accelerate it dramatically. So while you can’t stop the process entirely, the metabolic environment you create determines whether your thymus is 80% fat at 35 or still functional at 60.
2. The maintenance signal fades. A protein called Foxn1 keeps thymic epithelial cells alive and renewing. The gene for it doesn’t disappear with age. The body just stops activating it as strongly, likely because of the same inflammatory and metabolic shifts already described. When researchers experimentally boost Foxn1 in old animals, thymic function improves. The blueprint for a working thymus is still there. The metabolic environment just stops reading it.
3. Obesity accelerates all of it. Mice fed high-fat diets show dramatically faster thymic involution. The thymus merges into the surrounding body fat. Genetic obesity models produce the same result even on normal diets, so it’s the adiposity itself driving the damage, not just the dietary fat. Excess body fat doesn’t just sit there. It ages your immune system faster by accelerating the destruction of the organ that trains your immune cells.
The Bioenergetic Perspective: What Ray Peat Understood Decades Ago
Ray Peat was writing about the thymus as a metabolically dependent organ long before the 2023 NEJM study. His framework connects thymic health to the broader energetic state of the organism in ways new research increasingly supports.
Peat identified a specific list of factors that cause thymic atrophy: cortisol and other glucocorticoids, estrogen dominance, prostaglandins, polyunsaturated fatty acids, lipid peroxidation, nitric oxide, endotoxin, hypoglycemia, and ionizing radiation. He also identified what protects the thymus: progesterone and thyroid hormone.
Let’s look at each of these through the lens of current research.
Cortisol and the Stress Response
Hans Selye demonstrated in the mid-20th century that the thymus shrinks very early in the stress response. This is one of the most consistent findings in stress biology. Cortisol is directly toxic to thymocytes, inducing apoptosis. High cortisol also suppresses thymic epithelial cell function.
Chronically elevated cortisol, from sleep deprivation, overexercise, psychological stress, or metabolic dysfunction, creates a sustained suppressive environment for the thymus. Every time your cortisol stays elevated, your thymus takes a hit.
When Estrogen Goes Unopposed
Estrogen has a suppressive effect on thymic function, especially at higher levels or when unopposed by progesterone. It also stimulates the adrenal glands to produce more cortisol, amplifying the damage. At the same time, estrogen tends to shift the immune balance toward antibody production (B-cell activity) while suppressing aspects of thymus-dependent T-cell function.
This combination, weakened thymic control plus stimulated antibody production, is a recipe for autoimmune disease. The research backs this up. Autoimmune diseases are far more common in women, and conditions like lupus and rheumatoid arthritis fluctuate with estrogen levels.
There’s an interesting finding from menopause research. After menopause, when estrogen drops significantly, CT scans show women’s thymus scores actually increase briefly before continuing their decline. Ovariectomy in animal models produces a significant increase in thymus weight. This doesn’t mean estrogen is inherently harmful to the thymus. It means that the years of estrogen going unopposed by adequate progesterone, combined with the rising cortisol and declining thyroid function that typically accompany perimenopause, created a suppressive environment. When that burden lifts, the thymus partially rebounds.
Thyroid Hormone: The Thymus’s Best Friend
Research has demonstrated that T3 directly stimulates thymic epithelial cells to produce thymic hormones. Hypothyroid patients have lower thymulin levels. Treating hypothyroidism restores those levels.
Experimentally induced hypothyroidism in rats causes progressive thymic involution within four to seven weeks. The involution looks similar to aging: increased thymocyte death, increased macrophage activity consuming dying cells, structural deterioration.
This makes perfect sense if you think about it. Thyroid hormone drives mitochondrial respiration and energy production. The thymus is a metabolically demanding organ, constantly producing and destroying millions of cells. When cellular energy production drops, the thymus is one of the first organs to feel it. You can think of thymic involution as partly a bioenergetic problem: the cells of the thymus can’t maintain their function when energy availability declines.
Low thyroid function also tends to come packaged with elevated cortisol and elevated estrogen, both direct thymus suppressors. It’s a triple hit.
Progesterone: The Protective Hormone
R. Peat made a claim that stands out: “Progesterone is the only steroid hormone I know of which will cause the thymus to regenerate.”
This is a strong statement, but the mechanistic logic holds. Progesterone opposes estrogen’s effects on multiple levels. It reduces cortisol requirements by supporting adrenal function. It stabilizes cell membranes. It protects mitochondrial function. It inhibits inflammatory prostaglandin production. Every one of these actions would benefit the thymus.
Polyunsaturated Fatty Acids and Lipid Peroxidation
Thymic stromal cells lack hydrogen peroxide reductase and are particularly sensitive to oxidative damage from free radicals. PUFAs are highly susceptible to lipid peroxidation, which generates these free radicals.
Research shows that PUFAs suppress T-cell-mediated immune function and lymphocyte proliferation in both the spleen and thymus. This immunosuppression is attributed in part to increased lipid peroxidation and decreased antioxidant levels.
This connects to a broader principle: the accumulation of polyunsaturated fats in tissues with aging creates a pro-oxidative environment that damages metabolically active organs. The thymus, with its extremely high cell turnover, would be especially vulnerable.
Endotoxin and Hypoglycemia
Endotoxin (lipopolysaccharide from gut bacteria) activates inflammatory cascades that damage the thymus. A leaky gut, which becomes more common with aging and metabolic dysfunction, increases systemic endotoxin exposure.
Hypoglycemia triggers cortisol release to mobilize glucose, and cortisol damages the thymus. It also shifts metabolism away from glucose oxidation toward fat oxidation, reducing the efficient energy production the thymus depends on. Peat’s recommendation of adequate sugar in the diet as protection against metabolic aging has a direct thymus implication.
Supporting Your Thymus
Research points to several approaches that support thymic function and may slow further decline.
Metabolic Health Comes First
Obesity accelerates thymic aging more than almost any other factor. The connection runs through multiple mechanisms: increased inflammatory cytokines, elevated estrogen from aromatase activity in fat tissue, insulin resistance, and direct adipocyte infiltration of thymic tissue.
Maintaining healthy body composition protects the thymus.
Support Thyroid Function
Given the direct relationship between T3 and thymic hormone production, adequate thyroid function is essential. In practical terms, this means paying attention to the signs of subclinical hypothyroidism: cold hands and feet, low body temperature, fatigue, slow pulse. If your cells can’t produce energy efficiently, your thymus is suffering along with everything else.
Adequate dietary glucose is important because the liver requires glucose to convert T4 to the active T3. Protein is required for thyroid hormone transport proteins. Selenium and iodine are needed for thyroid hormone production.
Zinc
Zinc is the single most studied nutrient in relation to thymic function. Thymulin, the thymus’s signature hormone, is literally biologically inactive without zinc. Old mice given oral zinc supplementation for one to six months showed thymic regrowth, restoration of the epithelial cell network, disappearance of epithelial cysts, and partial recovery of peripheral immune function.
Zinc absorption declines with age, meaning older adults are more likely to be deficient precisely when their thymus needs it most. Good sources include oysters, red meat, eggs, and dairy. Plant sources contain zinc but phytates reduce absorption.
Chronic cortisol elevation is one of the most potent suppressors of thymic function. Adequate sleep, managing psychological stress, and maintaining stable blood sugar all help keep cortisol in a healthy range.
Progesterone opposes cortisol at multiple levels. For women, supporting progesterone production through adequate nutrition (especially cholesterol, vitamin A, and thyroid support) may indirectly protect the thymus.
Reduce PUFA Exposure
Based on the research showing PUFA-driven lipid peroxidation damages immune cells and suppresses lymphocyte proliferation in the thymus, reducing intake of polyunsaturated seed oils (soybean, corn, canola, safflower) and favoring saturated fats (butter, coconut oil) and monounsaturated fats (olive oil) could reduce oxidative stress on thymic tissue.
Vitamin E helps protect against PUFA peroxidation, but research suggests supplementation doesn’t fully prevent it. Reducing exposure is more effective than trying to neutralize the damage after the fact.
Vitamin A
Retinoic acid (the active form of vitamin A) directly regulates thymic epithelial cell development and maintenance. Vitamin A deficiency accelerates thymic involution. Good sources include liver, egg yolks, and dairy. Beta-carotene from plants is a poor substitute because conversion to retinol is inefficient and variable.
Reduce Endotoxin Exposure
Endotoxin is one of the most potent triggers of systemic inflammation. When the gut barrier is compromised, endotoxin leaks into the bloodstream and activates inflammatory cascades that damage the thymus directly. It also stimulates cortisol production, which compounds the damage.
Gut barrier integrity depends on practical things. Adequate protein, gelatin, broths supports the intestinal lining, which turns over rapidly and needs a constant supply of amino acids. Well-cooked vegetables and ripe fruits provide fiber without the irritation that raw or undercooked plant matter can cause.
Keeping digestion efficient matters too. Hypothyroidism slows gut motility, allowing bacteria to overgrow and increasing endotoxin production. Supporting thyroid function circles back here: faster transit time means less bacterial fermentation, less endotoxin, less inflammation, and less damage to the thymus.
Red and near-infrared light
Red and near-infrared light boost the energy-producing machinery inside your cells, specifically the final step of mitochondrial respiration. Since thymic decline is partly an energy problem improving mitochondrial efficiency could help the remaining thymic tissue stay functional longer.
A reasonable protocol for chest exposure: use a device that delivers both 660nm (red) and 850nm (near-infrared) wavelengths. Position yourself 6-12 inches from the panel. Start with 10 minutes per session, 3-5 times per week.
Thymic peptides and glandular extracts
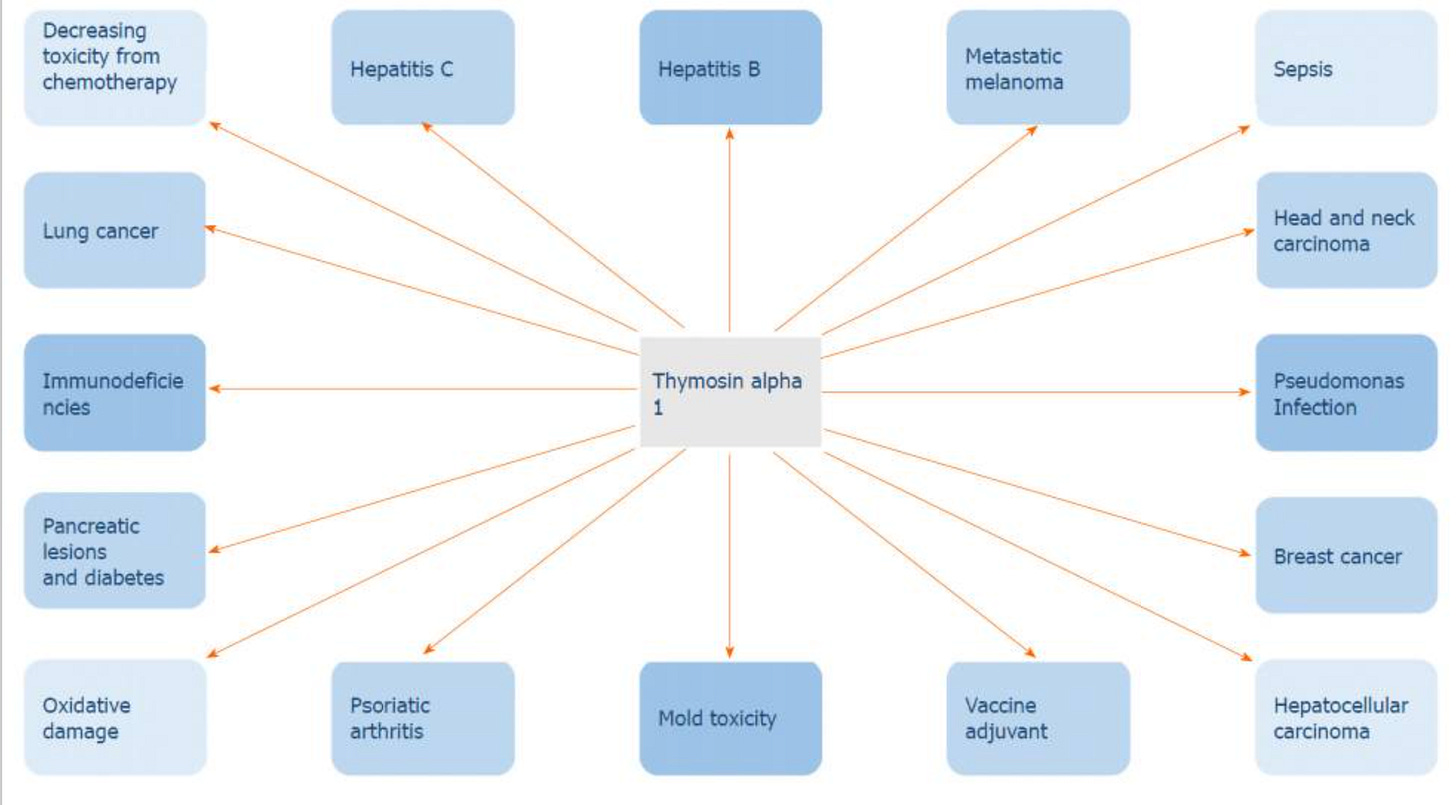
Thymic peptides like thymosin alpha-1, thymosin beta-4, thymulin, and thymopoietin are produced by thymic epithelial cells. They do more than just work inside the gland. They circulate in the blood and influence T-cell maturation, NK cell activity, dendritic cell function, and cytokine balance. Their activity is tied to cortisol levels, so they’re part of the conversation between your immune system and your stress response.
Thymosin alpha-1 has the strongest evidence behind it. Its synthetic form (thymalfasin) has FDA orphan drug designation for melanoma, hepatitis B, DiGeorge syndrome, and hepatocellular carcinoma. It’s been studied in HIV, sepsis, and COVID-19 with mixed but often promising results. In Europe, glandular thymus extracts containing various combinations of these peptides have been used clinically for decades in oncology and chronic immune dysfunction. From 2020 study “Thymosin alpha 1 has exhibited the ability to restrain tumor growth, hence its use in the treatment of various cancers.”
Zinc-thymulin complexes are worth knowing about too. Thymulin is biologically inactive without zinc, so supplementing both together targets the exact deficit that develops when aging or poor zinc status shuts down thymulin function. Synthetic zinc-thymulin complexes exist as injectable peptides available through specialized clinics, but they’re not widely accessible as standard supplements. For most people, ensuring sufficient zinc intake (through food or supplementation) is the practical way to support whatever thymulin their thymus is still producing.
None of this replaces a functioning thymus. But for people dealing with immune suppression from chemotherapy, radiation, chronic infections, or significant immune decline, these are real compounds with real mechanisms behind them.
***********************************************
The thymus sits at the intersection of metabolism, immunity, and aging in a way that few other organs do. Its decline tracks almost perfectly with the rise of age-related disease: cancer, autoimmunity, increased infection susceptibility, chronic inflammation.
Standard medicine has treated these as separate problems requiring separate solutions: chemotherapy for cancer, immunosuppressants for autoimmunity, vaccines for infection risk, anti-inflammatory drugs for chronic inflammation. But the thymus offers a unifying lens. If the organ responsible for immune education and surveillance is deteriorating, you’d expect exactly this constellation of problems. Thymus doesn’t shrink in isolation. It shrinks when the metabolic environment deteriorates, when cortisol is chronically elevated, when estrogen is unopposed, when thyroid function is insufficient, when lipid peroxidation is running unchecked, when the body is overloaded with inflammatory signals from a compromised gut.
Fix the metabolic environment, and the thymus may have more room to function.
The Greeks called the thymus the seat of the soul. They may have been closer to the truth than the surgeons who casually discarded it on the operating table.
Your thymus is still there. It’s still working, and it deserves your attention. Now.
References:
1. Kooshesh KA, Foy BH, Sykes DB, Gustafsson K, Scadden DT. Health Consequences of Thymus Removal in Adults. New England Journal of Medicine. 2023
2. Kooshesh KA, Foy BH, Baylis RA, Gustafsson K, Zlotoff DA, Neilan TG, Scadden DT. Adult thymectomy is associated with increased mortality risk from cancer but not cardiovascular disease. Blood Advances. 2025
3. Liang Z, Dong X, Zhang Z, Zhang Q, Zhao Y. Age-related thymic involution: Mechanisms and functional impact. Aging Cell. 2022
4. Dixit VD. Thymic fatness and approaches to enhance thymopoietic fitness in aging. Current Opinion in Immunology. 2010
5. Yang H, Youm YH, Vandanmagsar B, Rber A, Stephens JM, Dixit VD. Obesity accelerates thymic aging. 2009
6. Mocchegiani E, Santarelli L, Muzzioli M, Fabris N. Reversibility of the thymic involution and of age-related peripheral immune dysfunctions by zinc supplementation in old mice. International Journal of Immunopharmacology. 1995
7. Dardenne M, Savino W, Borrih S, Bach JF. A zinc-dependent epitope on the molecule of thymulin, a thymic hormone. Proceedings of the National Academy of Sciences. 1985
8. Savino W, Dardenne M. Nutritional imbalances and infections affect the thymus: consequences on T-cell-mediated immune responses. Proceedings of the Nutrition Society. 2010
9. Dooley J, Liston A. Molecular control over thymic involution: From cytokines and microRNA to aging and adipose tissue. European Journal of Immunology. 2012
10. Min H, Montecino-Rodriguez E, Dorshkind K. Reassessing the role of growth hormone and sex steroids in thymic involution. Clinical Immunology. 2006
11. AbouRabia N, Kendall MD. Involution of the rat thymus in experimentally induced hypothyroidism. Cell and Tissue Research. 1994
12. Fabris N, Mocchegiani E, Provinciali M. Thyroid-thymus interactions during development and aging. Hormone Research. 1989
13. Sauce D, Larsen M, Fastenackels S, et al. Evidence of premature immune aging in patients thymectomized during early childhood. Journal of Clinical Investigation. 2009
14. Dominari A, Hathaway D, Pandav K, et al. Thymosin alpha 1: A comprehensive review of the literature. World Journal of Virology. 2020
15. Skotnicki AB. Therapeutic application of calf thymus extract (TFX). Medical Oncology and Tumor Pharmacotherapy. 1989
16. Peat R. Immunodeficiency, dioxins, stress, and the hormones.
17. Peat R. Aging, estrogen, and progesterone.
18. Selye H. The Stress of Life. McGraw-Hill. 1956.



I eat 2 oysters most mornings after doing a mitochondrial analysis thru Chris Maserjohn. Increased liver consumption, too, but not every day. Not sure how it's affecting my thymus, but I feel overall stronger, stronger than I did even 20 years ago. I'm 54 now. Thank you for the in depth content, it is much appreciated.
New info. And much to ponder. Thank you.